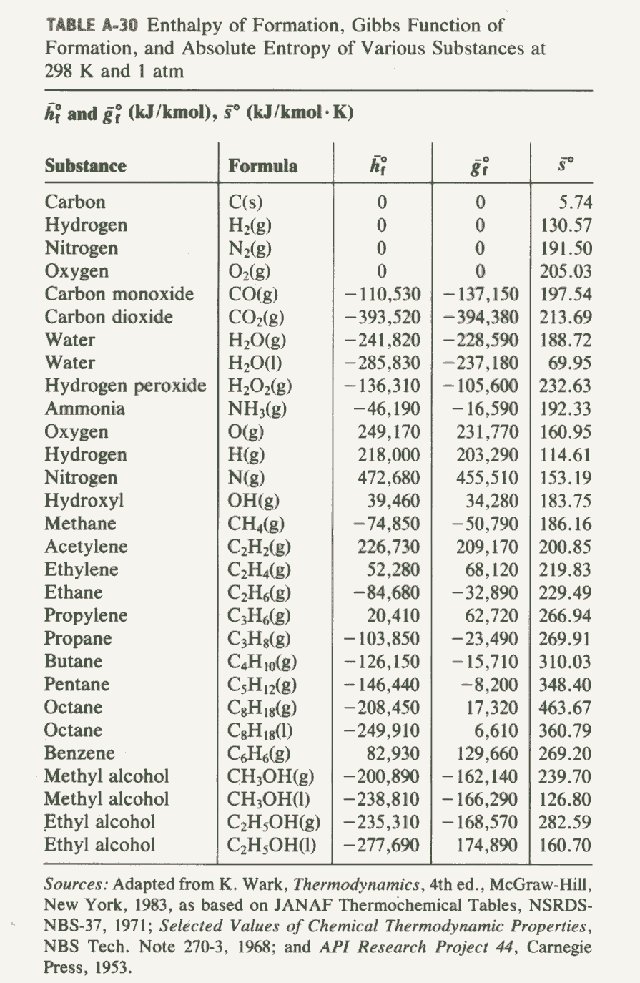
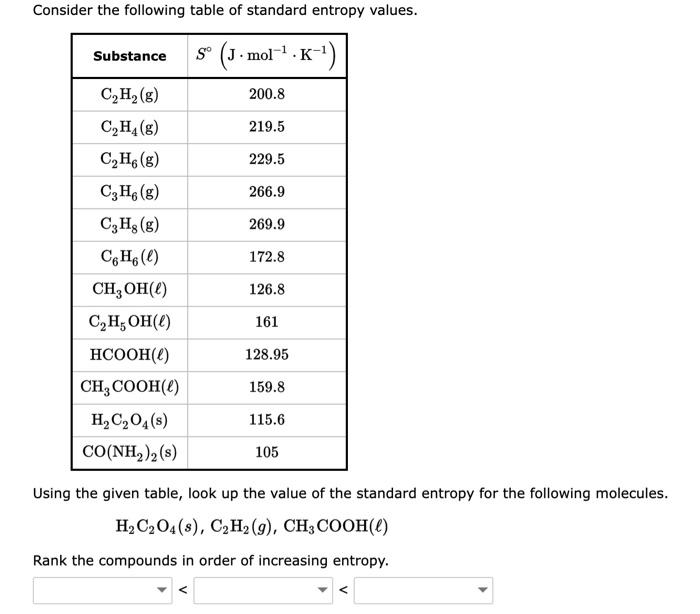
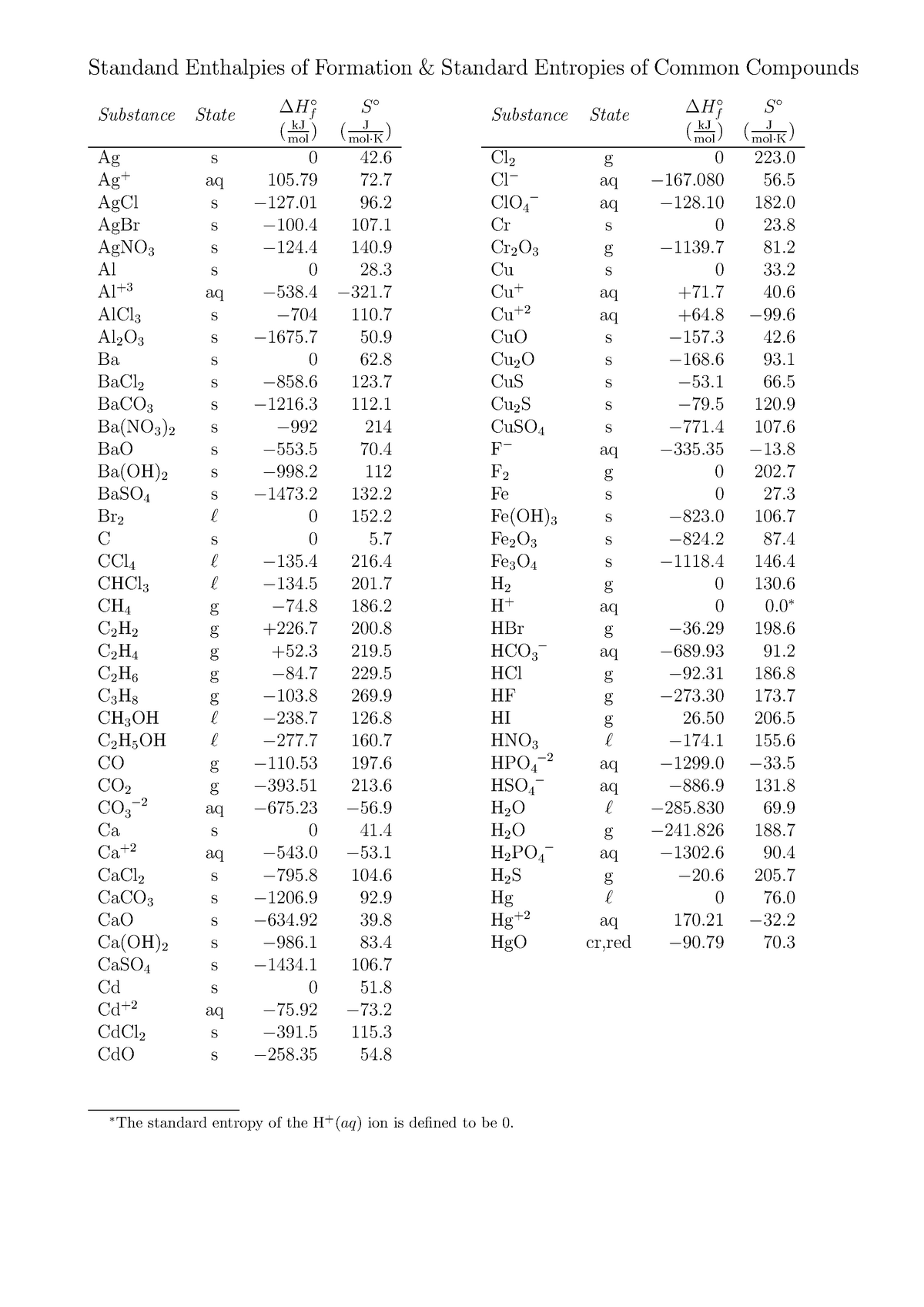
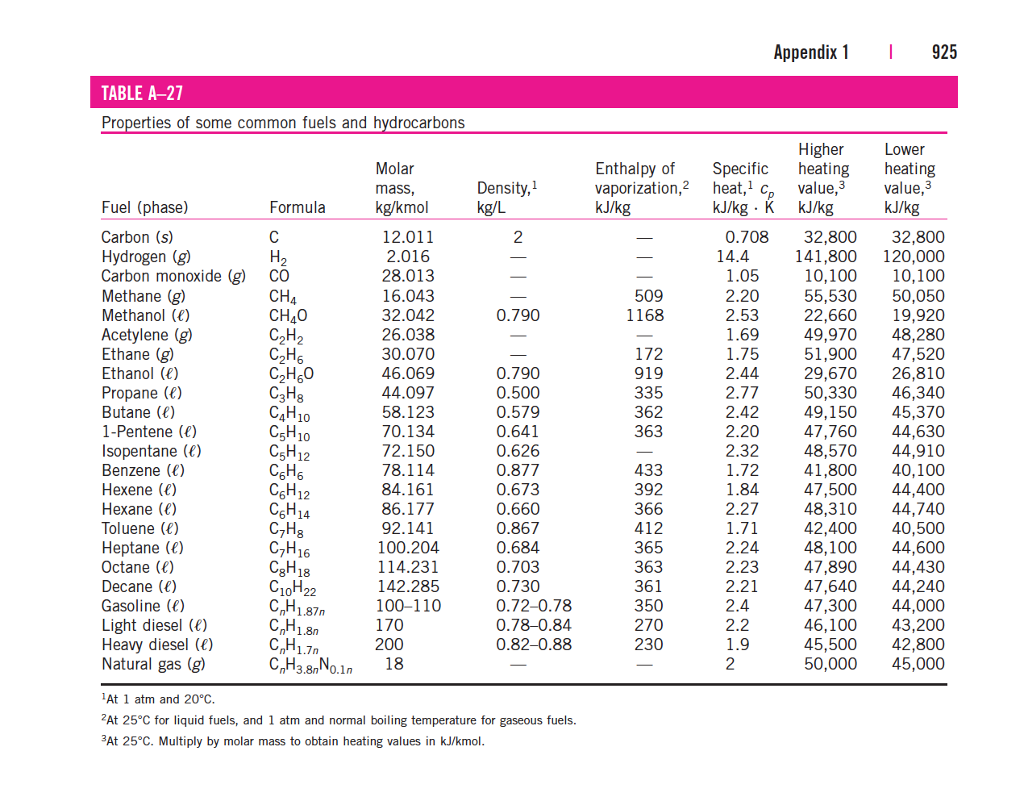
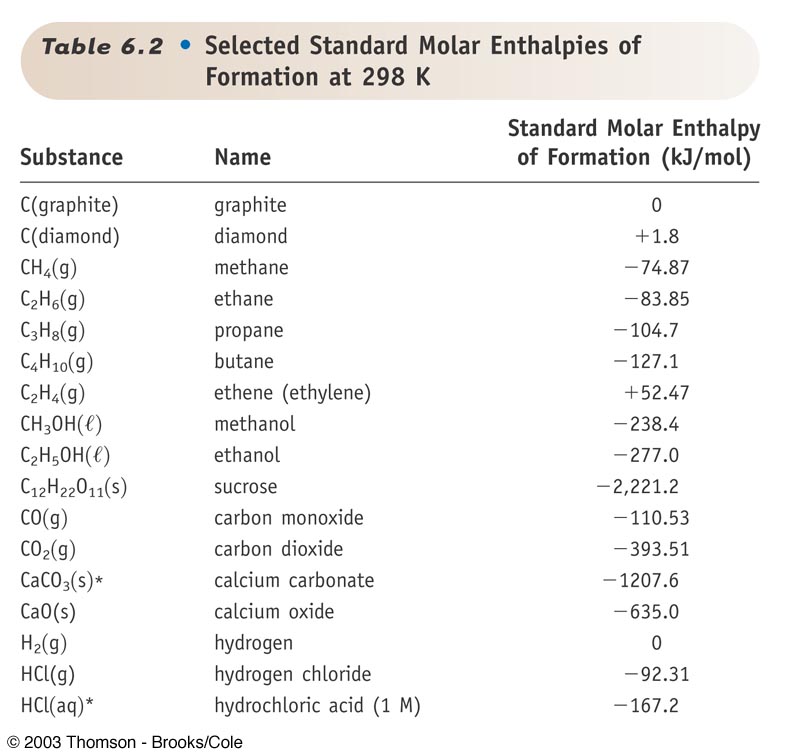
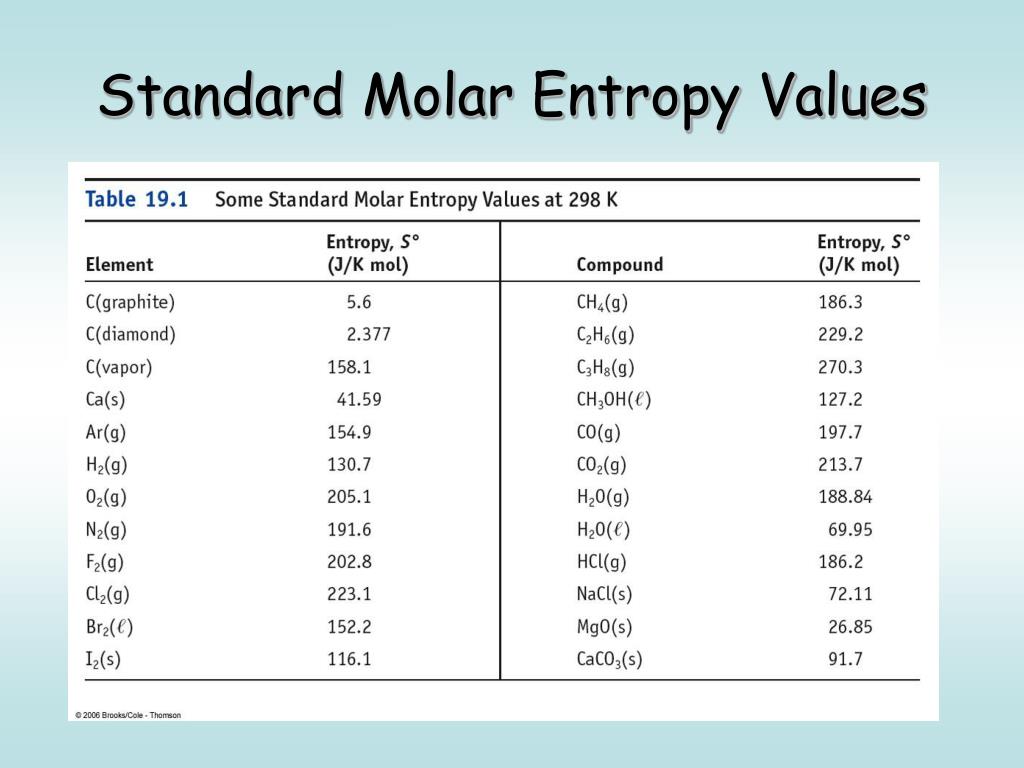
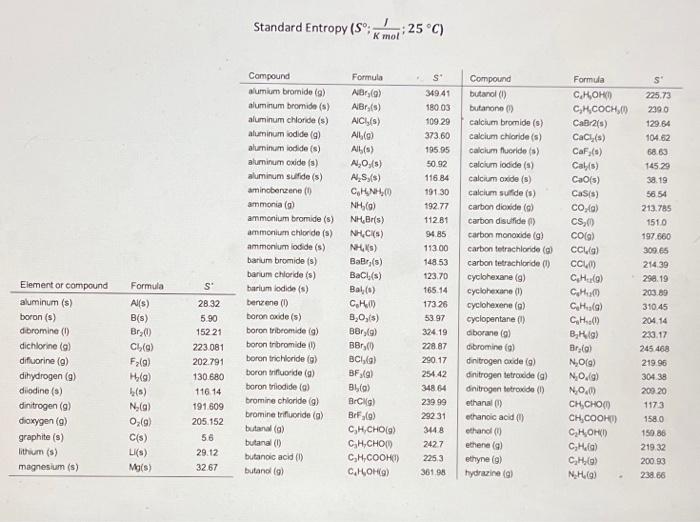
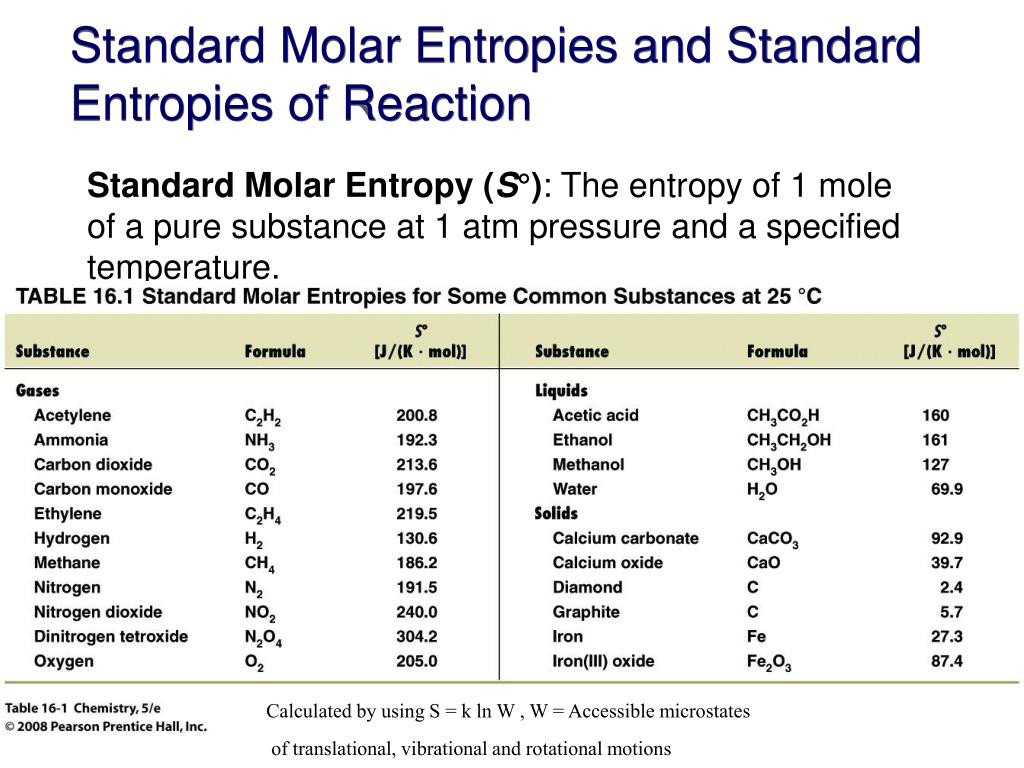
Standard Entropy Of Formation Table
Standard Entropy Of Formation Table - 151 rows internationally agreed, internally consistent, values for the thermodynamic. The table below shows the standard enthalpy of formation, the standard gibbs free energy of. This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds. Values of the standard molar entropies of various substances at 298 k (25°c). ∗the standard entropy of the h+(aq) ion is defined to be 0. Definition and explanation of the terms standard state and standard enthalpy of formation, with. 100 rows standard heats and free energies of formation and absolute entropies of elements.
Definition and explanation of the terms standard state and standard enthalpy of formation, with. The table below shows the standard enthalpy of formation, the standard gibbs free energy of. ∗the standard entropy of the h+(aq) ion is defined to be 0. Values of the standard molar entropies of various substances at 298 k (25°c). 151 rows internationally agreed, internally consistent, values for the thermodynamic. This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds. 100 rows standard heats and free energies of formation and absolute entropies of elements.
Definition and explanation of the terms standard state and standard enthalpy of formation, with. Values of the standard molar entropies of various substances at 298 k (25°c). ∗the standard entropy of the h+(aq) ion is defined to be 0. 151 rows internationally agreed, internally consistent, values for the thermodynamic. This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds. 100 rows standard heats and free energies of formation and absolute entropies of elements. The table below shows the standard enthalpy of formation, the standard gibbs free energy of.
Standard Enthalpies of Formation SchoolWorkHelper
100 rows standard heats and free energies of formation and absolute entropies of elements. The table below shows the standard enthalpy of formation, the standard gibbs free energy of. This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds. Values of the standard molar entropies of various substances at 298 k (25°c). 151 rows internationally.
Solved Consider the following table of standard entropy
This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds. 151 rows internationally agreed, internally consistent, values for the thermodynamic. Definition and explanation of the terms standard state and standard enthalpy of formation, with. 100 rows standard heats and free energies of formation and absolute entropies of elements. The table below shows the standard enthalpy.
Standard entropy table Lasialabama
This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds. Values of the standard molar entropies of various substances at 298 k (25°c). ∗the standard entropy of the h+(aq) ion is defined to be 0. 100 rows standard heats and free energies of formation and absolute entropies of elements. The table below shows the standard.
TABLE A286 Enthalpy of formation, Gibbs function of
Definition and explanation of the terms standard state and standard enthalpy of formation, with. Values of the standard molar entropies of various substances at 298 k (25°c). The table below shows the standard enthalpy of formation, the standard gibbs free energy of. 151 rows internationally agreed, internally consistent, values for the thermodynamic. 100 rows standard heats and free energies of.
to Adobe GoLive 6
This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds. Definition and explanation of the terms standard state and standard enthalpy of formation, with. 151 rows internationally agreed, internally consistent, values for the thermodynamic. Values of the standard molar entropies of various substances at 298 k (25°c). The table below shows the standard enthalpy of.
Standard entropy table Lasialabama
The table below shows the standard enthalpy of formation, the standard gibbs free energy of. 151 rows internationally agreed, internally consistent, values for the thermodynamic. 100 rows standard heats and free energies of formation and absolute entropies of elements. Values of the standard molar entropies of various substances at 298 k (25°c). ∗the standard entropy of the h+(aq) ion is.
Standard entropy table filmqlero
Definition and explanation of the terms standard state and standard enthalpy of formation, with. ∗the standard entropy of the h+(aq) ion is defined to be 0. This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds. Values of the standard molar entropies of various substances at 298 k (25°c). The table below shows the standard.
Solved 3. Use the tables of standard enthalpy of formation
The table below shows the standard enthalpy of formation, the standard gibbs free energy of. 100 rows standard heats and free energies of formation and absolute entropies of elements. Values of the standard molar entropies of various substances at 298 k (25°c). Definition and explanation of the terms standard state and standard enthalpy of formation, with. This table lists the.
Standard entropy wsmzaer
This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds. The table below shows the standard enthalpy of formation, the standard gibbs free energy of. ∗the standard entropy of the h+(aq) ion is defined to be 0. 151 rows internationally agreed, internally consistent, values for the thermodynamic. 100 rows standard heats and free energies of.
[Solved] Calculate the standard molar entropy change for the formation
100 rows standard heats and free energies of formation and absolute entropies of elements. Values of the standard molar entropies of various substances at 298 k (25°c). The table below shows the standard enthalpy of formation, the standard gibbs free energy of. This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds. 151 rows internationally.
The Table Below Shows The Standard Enthalpy Of Formation, The Standard Gibbs Free Energy Of.
This table lists the standard enthalpies (δh°), the free energies (δg°) of formation of compounds. 100 rows standard heats and free energies of formation and absolute entropies of elements. Values of the standard molar entropies of various substances at 298 k (25°c). Definition and explanation of the terms standard state and standard enthalpy of formation, with.
∗The Standard Entropy Of The H+(Aq) Ion Is Defined To Be 0.
151 rows internationally agreed, internally consistent, values for the thermodynamic.